- #Iso 17025 2017 mandatory documents how to#
- #Iso 17025 2017 mandatory documents manual#
- #Iso 17025 2017 mandatory documents upgrade#
- #Iso 17025 2017 mandatory documents iso#
#Iso 17025 2017 mandatory documents iso#

#Iso 17025 2017 mandatory documents manual#
ISO 17025:2017 Manual (8 Chapters and 1 Annexure): A sample ISO/IEC 17025 manual with a quality policy that meets the requirement of standard accreditation.It increases your team's confidence and accelerates organization towards achieving ISO certification with proper implementation and getting the benefits of continuous improvement.The contents of the document kit which we offer include more than 100 document files as listed below:
#Iso 17025 2017 mandatory documents how to#
Readymade documents guide you on how to write quality manual and procedures for each department. It helps to review macro-level survey of the implemented system for how each requirements of standard is implemented in the testing or calibration laboratory. Our ISO/IEC 17025:2017 Training ppt presentation will guide users to learn requirements of Laboratory accreditation system and ISO/IEC 17025:2017 standard in day-to-day process. Such ISO 17025 documents are designed under guidance of experienced ISO consultant, and therefore, reduce repetitive work and provide accurate documented information.įor preparing Documents based on ISO/IEC 17025:2017 for your laboratory, write us at 17025:2017 Auditor Training - PPT Presentation The key benefit of using such readymade documents is that they save time and cost in the process of documentation, and hence, certification. The complete set of readymade documents applies to both small and large size laboratories.
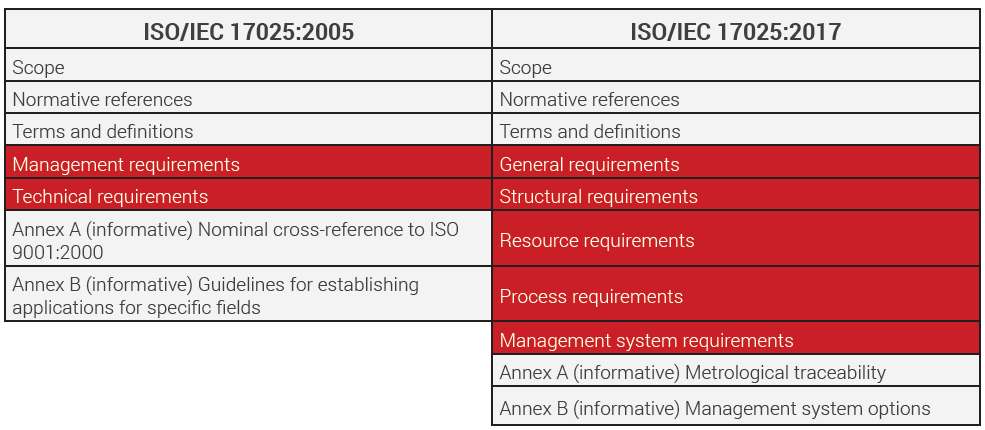
The ready-to-use ISO/IEC 17025:2017 documents for testing and calibration laboratories include sample copy of quality manual drafted and endorsed to ensure effective laboratory management system, mandatory procedures, SOPs, sample formats as well as ready-to-use ISO 17025 audit checklists, etc., in editable word formats.

ISO 17025:2017 Documentation - Manual, Procedures, Checklistĭ has joined hands with Global Manager Group, world's leading ISO consultant, to offer ISO/IEC 17025 Documentation Toolkit, which is designed as per latest version of standard requirements. We also provide them online training that helps them to learn ISO/IEC 17025:2017 document requirements, and saves their lots of time in documentation. We are offering online ISO 17025:2017 documentation consultancy, in which we collect required information from clients via e-mail and other digital resources and prepare documents accordingly by using our documentation toolkit. To achieve ISO/IEC 17025:2017 certification, organizations must have to implement quality management system in their calibration/testing laboratories and prepare documents that define the implemented system.
#Iso 17025 2017 mandatory documents upgrade#
The new version of ISO/IEC 17025 was released in 2017 by the ISO and laboratories have 3 years' time to upgrade their existing system. The ISO/IEC 17025:2017 is a revised standard for accreditation of testing and calibration laboratories.
